Press release

NOBELFÖRSAMLINGEN KAROLINSKA INSTITUTET
THE NOBEL ASSEMBLY AT THE KAROLINSKA INSTITUTE
The Nobel Assembly at the Karolinska Institute has today decided to award the Nobel Prize in Physiology or Medicine for 1986 jointly to
Stanley Cohen and Rita Levi-Montalcini
for their discoveries of “growth factors”.
Summary
The Nobel Prize in Physiology or Medicine is awarded for discoveries which are of fundamental importance for our understanding of the mechanisms which regulate cell and organ growth. The pattern of cellular growth has long been known, but it is the Italian developmental biologist Rita Levi-Montalcini and the American biochemist Stanley Cohen with their discovery of nerve growth factor (NGF) and epidermal growth factor (EGF), respectively, who could show how the growth and differentiation of a cell is regulated. NGF and EGF were the first of many growth-regulating signal substances to be discovered and characterized.
The discovery of NGF and EGF has opened new fields of widespread importance to basic science. As a direct consequence we may increase our understanding of many disease states such as developmental malformations, degenerative changes in senile dementia, delayed wound healing and tumour diseases. The characterization of these growth factors is therefore expected, in the near future, to result in the development of new therapeutic agents and improved treatment in various clinical diseases.
The Regulation of Growth
Adult man consists of billions of cells. However, he begins from a single cell which contains the genetic material coding for the complete individual. The first cell divides, and in the beginning, the daughter cells are identical. Soon, however, the cells begin to exhibit slightly different characteristics. This unique specialization of cells is termed differentiation. The pattern for growth and differentiation has long been established but the regulation of this development has remained unknown. It was only during the past 2-3 decades with the discovery of growth factors that our understanding of the regulation of cell’s growth and differentiation has begun to be clarified.
Today, we know that cells communicate with each other via signal substances, hormones. Initially it was believed that hormones were only produced in specialized glands such as the pituitary, from which hormones such as growth hormone, were released into the blood stream. It has now become clear that many cell types synthesize signal substances or hormones which have their effects both on their cell of origin as well as on neighbouring cells. By this mechanism, cells can influence the development of their neighbours.
The scientists in the 1940’s and 1950’s already knew that the addition of blood or organ extracts to cells in culture resulted in their successful growth. They did not know, however, the identity of the active substances, just as cancer researchers understood little of the unregulated growth of tumour cells.
The Discovery of NGF
The discovery of nerve growth factor (NGF) in the beginning of the 1950’s is a fascinating example of how a skilled observer can create a concept out of apparent chaos. Until this time, experimental neurobiologists did not understand how the development of the nervous system was regulated to result in the final complete innervation of the body. The investigation of NGF’s role in the development of the nervous system, as well as later, in adult neural function, has been a lifelong dedication for Rita Levi-Montalcini. Developmental biologist Rita Levi-Montalcini, who in the beginning of 1950’s moved from her homeland Italy, to Viktor Hamburger’s laboratory in St. Louis, USA, showed in 1952 that when tumours from mice were transplanted to chick embryos they induced potent growth of the chick embryo nervous system, specifically sensory and sympathetic nerves. Since this outgrowth did not require direct contact between the tumour and the chick embryo, Rita Levi-Montalcini concluded that the tumour released a nerve growth-promoting factor which had a selective action on certain types of nerves. Following this discovery, Rita Levi-Montalcini turned to a more sensitive cell culture system in order to measure NGF activity in various extracts. NGF proved to be an extremely potent biological substance. A sensory or sympathetic nerve cell reacted within 30 seconds to the addition of minute quantities of NGF. One billionth part of a gram of NGF per ml of culture medium exerted a potent growth-promoting effect. A few minutes after the addition of NGF, nerve fibres began to grow out from the ganglion which after a day’s exposure to NGF resembled a sun surrounded by rays (Figure 1). This biological assay to detect NGF paved the way for the next step in this pathway of discovery – identification of the active nerve growth-promoting substance.
 |
Figure 1. The classical biological assay for the measurement of NGF which was developed by Rita Levi-Montalcini. Sensory ganglion dissected from chick embryo is cultured in the presence of extract to be measured. Nerve fibre outgrowth from the chick ganglion is determined after 24 hours. The lowest concentration of the extract which causes a halo of nerve fibre outgrowth (right hand side figure) contains 1 biological unit of NGF. This is equivalent to a concentration of approximately 10 nanograms NGF/ml culture medium (10 ng=1/100,000 of 1 milligram). The left hand side ganglion has been incubated without NGF being present in the medium and is in the process of dying.
The figure has been published in Scientific American 1979, 240, p. 48.
The Characterization of NGF
In 1953, biochemist Stanley Cohen, joined the research group in St. Louis. Three years later they had purified a nerve growth-promoting extract from mouse tumour which contained both protein and nucleic acids. To determine which of these components was active, Stanley Cohen added snake venom containing a high concentration of a nucleic acid-degrading enzyme. To his surprise, the snake venom contained more nerve growth-promoting activity than the tumour itself. When added alone to the incubation medium, the snake venom induced an enormous outgrowth of sympathetic nerve fibres. The group followed up this unexpected finding by systematically searching for the presence of NGF in various tissues. In 1958, they discovered another rich source of NGF – a salivary gland in the male mouse.
With the help of snake venom and salivary gland extract, Stanley Cohen was now able to purify NGF and produce antibodies against NGF. Just as NGF induced nerve growth, so did the addition of the NGF antibodies to the incubation medium inhibit it. The chemical structure of NGF is now known to consist of 118 amino acids. Two such chains join together to build a biologically active molecule.
The advances were a milestone in developmental neurobiology. For the first time, chemically defined signal substances could be used in the search for mechanisms which regulated neural development.
NGF is Necessary for the Survival of Nerve Cells
NGF is found in mammals, birds, reptiles, amphibians and fishes. Many cell types in these species synthesize and release NGF during development. The growth of nerve fibres is stimulated via this mechanism. Nerve fibres grow towards the source of NGF which is taken up in the fibre ends and transported back to the nerve cell body. One can imagine that tissues lure nerve fibres to them by sending out NGF. An excess of sensory and sympathetic nerve cells is produced during development. Only those which establish contact with a target organ producing NGF survive. With modern gene technology it has now been possible to characterize the NGF gene in man and animals. Hybridization techniques identifying the messenger RNA for NGF have mapped the tissues that synthesize NGF. Gene technology also makes possible recombinant NGF, which opens the way for its future application in clinical medicine.
The Discovery of EGF
During the course of his studies of NGF Stanley Cohen observed an unexpected acceleration of development when he injected salivary gland extract to newborn mice. The mice displayed precocious eyelid opening and tooth eruption. The explanation was that the salivary gland extract contained another growth factor apart from NGF. Cohen termed this substance epidermal growth factor (EGF) because it could stimulate the proliferation of epithelial cells in skin and cornea. He raised antibodies against EGF as he previously had against NGF.
In the following years Cohen purified and determined the amino acid sequence of EGF (Figure 2). For the first time scientists had a factor available which stimulated epithelial cell growth and allowed studies of the growth process. Cohen and his coworkers found that EGF enhanced a cascade of events including stimulation of glucose and amino acid transport, activation of protein synthesis and initiation of DNA synthesis and cell replication. In later studies both Cohen and others have shown that EGF stimulates the growth of a large variety of cells including fibroblasts, liver cells, and vascular cells as well as endocrine cells the thyroid, ovaries and pituitary glands.
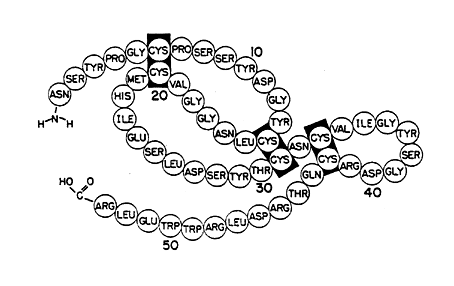
Figure 2. The amino acid sequence of EGF with placement of disulfide bonds.
The figure has been published in J. Biol. Chem. 1973, 248, p. 7670.
The presence of specific binding sites, termed receptors, on the surface of target cells was a prerequisite for the action of EGF. These receptors catch EGF and the EGF-receptor complex is taken into the cell. One important step in the events leading to the biological action of EGF is the phosphorylation of a specific amino acid tyrosine on the EGF receptor.
The finding of tyrosine-specific autophosphorylation of the receptor was a breakthrough in our understanding of how a signal from the outside reaches the inside of the cell. It has later been shown that this event is a general pathway through which many growth factors mediate their effects.
The knowledge of the regulation of normal cell growth has provided new insights into cell transformation and tumour growth. Studies on certain virus-induced tumours have led to the discoveries of special genes, called oncogenes, which play a role in the transformation of cells. Amongst these oncogenes there is one, which codes for the synthesis of a protein with homology to the EGF-receptor. Another oncogene product shows similarity with a later discovered growth factor derived from the blood platelets (PDGF). As often occurs, increased knowledge of normal events has led to an improved understanding of disease.
Search for Other Growth Factors
During the last decade, several growth factors have been isolated and characterized by different research groups. For example, somatomedin or insulin-like growth factor, which mediates the growth-promoting effect of growth hormone, was isolated from human plasma. Platelet derived growth factor (PDGF) was isolated and shown to stimulate growth of mesenchymal cells. Interleukin-2 was isolated and shown to promote growth of the lymphocytes of the immune system. From tumours, endothelial cell growth factor (ECGF) was isolated and shown to have similarities to the fibroblast growth factor (FGF).
All research groups who discovered “new” growth factors have followed in the tracks of Levi-Montalcini and Cohen. In the research area of growth factors and their biological action, Levi-Montalcini and Cohen have created a scientific school with an increasing number of followers.
Clinical Application of NGF and EGF
Clarification of the mechanisms regulating the growth and survival of cells as well as their differentiation are of great interest to basic science. However, this knowledge can be expected to improve our understanding of the pathogenesis of several clinical disorders such as malformation and errors of development, degenerative changes such as occur in senile dementia, delayed wound healing, muscle dystrophy as well as tumour diseases.
An important future field of NGF therapy is the possibility of enhancing the reparative process after damage to nerves in the periphery as well as in the central nervous system. The recent finding of NGF in the brain has raised great expectation. An important pathway in the brain with acetylcholine as a transmittor substance seems to be sensitive to NGF.
In studies performed on animals, EGF has been shown to enhance the healing of wounds in both skin and cornea. Limited amounts of human EGF have restricted its use in humans. Only now that it is possible to produce recombinant EGF have clinical trials been started. A future application of EGF to enhance the repair of wounds in skin and cornea by, for example, local application after damage or surgery can be anticipated. Autotransplantation of skin would be improved if epithelial cells could be quickly cultivated outside the body with the help of EGF. It would also be possible that antagonists to EGF or antibodies to the EGF-receptors on the cell surface could be useful in the treatment of tumours in which derangement of EGF or the EGF-receptor is involved in their transformation.
References
Rita Levi-Montalcini & Pietro Calissano: The Nerve-Growth Factor. Scientific American 1979, 240, pp. 44-53.
B Alberts, D Bray, J Lewis, M Raff, K Roberts & J D Watson, Eds.: Molecular Biology of the Cell. Garland Publ. Incorp., New York and London, 1983.
Nobel Prizes and laureates
Six prizes were awarded for achievements that have conferred the greatest benefit to humankind. The 14 laureates' work and discoveries range from quantum tunnelling to promoting democratic rights.
See them all presented here.
