Press release
English
French
German
Swedish

7 October 2002
The Nobel Assembly at Karolinska Institutet has today decided to award
The Nobel Prize in Physiology or Medicine for 2002 jointly to
Sydney Brenner, H. Robert Horvitz and John E. Sulston
for their discoveries concerning “genetic regulation of organ development and programmed cell death”
Summary
The human body consists of hundreds of cell types, all originating from the fertilized egg. During the embryonic and foetal periods, the number of cells increase dramatically. The cells mature and become specialized to form the various tissues and organs of the body. Large numbers of cells are formed also in the adult body. In parallel with this generation of new cells, cell death is a normal process, both in the foetus and adult, to maintain the appropriate number of cells in the tissues. This delicate, controlled elimination of cells is called programmed cell death.
This year’s Nobel Laureates in Physiology or Medicine have made seminal discoveries concerning the genetic regulation of organ development and programmed cell death. By establishing and using the nematode Caenorhabditis elegans as an experimental model system, possibilities were opened to follow cell division and differentiation from the fertilized egg to the adult. The Laureates have identified key genes regulating organ development and programmed cell death and have shown that corresponding genes exist in higher species, including man. The discoveries are important for medical research and have shed new light on the pathogenesis of many diseases.
Sydney Brenner (b 1927), Berkeley, CA, USA, established C. elegans as a novel experimental model organism. This provided a unique opportunity to link genetic analysis to cell division, differentiation and organ development – and to follow these processes under the microscope. Brenner’s discoveries, carried out in Cambridge, UK, laid the foundation for this year’s Prize.
John Sulston (b 1942), Cambridge, England, mapped a cell lineage where every cell division and differentiation could be followed in the development of a tissue in C. elegans. He showed that specific cells undergo programmed cell death as an integral part of the normal differentiation process, and he identified the first mutation of a gene participating in the cell death process.
Robert Horvitz (b 1947), Cambridge, MA, USA, has discovered and characterized key genes controlling cell death in C. elegans. He has shown how these genes interact with each other in the cell death process and that corresponding genes exist in humans.
Cell lineage – from egg to adult
All cells in our body are descendents from the fertilized egg cell. Their relationship can be referred to as a cellular pedigree or cell lineage. Cells differentiate and specialize to form various tissues and organs, for example muscle, blood, heart and the nervous system. The human body consists of several hundreds of cell types, and the cooperation between specialized cells makes the body function as an integrated unit. To maintain the appropriate number of cells in the tissues, a fine-tuned balance between cell division and cell death is required. Cells have to differentiate in a correct manner and at the right time during development in order to generate the correct cell type.
It is of considerable biological and medical importance to understand how these complicated processes are controlled. In unicellular model organisms, e.g. bacteria and yeast, organ development and the interplay between different cells cannot be studied. Mammals, on the other hand, are too complex for these basic studies, as they are composed of an enormous number of cells. The nematode C. elegans, being multi-cellular, yet relatively simple, was therefore chosen as the most appropriate model system, which has then led to characterization of these processes also in humans.
Programmed cell death
Normal life requires cell division to generate new cells but also the presence of cell death, so that a balance is maintained in our organs. In an adult human being, more than a thousand billion cells are created every day. At the same time, an equal number of cells die through a controlled “suicide process”, referred to as programmed cell death.
Developmental biologists first described programmed cell death. They noted that cell death was necessary for embryonic development, for example when tadpoles undergo metamorphosis to become adult frogs. In the human foetus, the interdigital mesoderm initially formed between fingers and toes is removed by programmed cell death. The vast excess of neuronal cells present during the early stages of brain development is also eliminated by the same mechanism.
The seminal breakthrough in our understanding of programmed cell death was made by this year’s Nobel Laureates. They discovered that specific genes control the cellular death program in the nematode C. elegans. Detailed studies in this simple model organism demonstrated that 131 of totally 1090 cells die reproducibly during development, and that this natural cell death is controlled by a unique set of genes.
The model organism C. elegans
Sydney Brenner realized, in the early 1960s, that fundamental questions regarding cell differentiation and organ development were hard to tackle in higher animals. Therefore, a genetically amenable and multicellular model organism simpler than mammals, was required. The ideal solution proved to be the nematode Caenorhabditis elegans. This worm, approximately 1 mm long, has a short generation time and is transparent, which made it possible to follow cell division directly under the microscope.
Brenner provided the basis in a publication from 1974, in which he broke new ground by demonstrating that specific gene mutations could be induced in the genome of C. elegans by the chemical compound EMS (ethyl methane sulphonate). Different mutations could be linked to specific genes and to specific effects on organ development. This combination of genetic analysis and visualization of cell divisions observed under the microscope initiated the discoveries that are awarded by this year’s Nobel Prize.
Mapping the cell lineage
John Sulston extended Brenner’s work with C. elegans and developed techniques to study all cell divisions in the nematode, from the fertilized egg to the 959 cells in the adult organism. In a publication from 1976, Sulston described the cell lineage for a part of the developing nervous system. He showed that the cell lineage is invariant, i.e. every nematode underwent exactly the same program of cell division and differentiation.
As a result of these findings Sulston made the seminal discovery that specific cells in the cell lineage always die through programmed cell death and that this could be monitored in the living organism. He described the visible steps in the cellular death process and demonstrated the first mutations of genes participating in programmed cell death, including the nuc-1 gene. Sulston also showed that the protein encoded by the nuc-1 gene is required for degradation of the DNA of the dead cell.
Identification of “death genes”
Robert Horvitz continued Brenner’s and Sulston’s work on the genetics and cell lineage of C. elegans. In a series of elegant experiments that started during the 1970s, Horvitz used C. elegans to investigate whether there was a genetic program controlling cell death. In a pioneering publication from 1986, he identified the first two bona fide “death genes”, ced-3 and ced-4. He showed that functional ced-3 and ced-4 genes were a prerequisite for cell death to be executed.
Later, Horvitz showed that another gene, ced-9, protects against cell death by interacting with ced-4 and ced-3. He also identified a number of genes that direct how the dead cell is eliminated. Horvitz showed that the human genome contains a ced-3-like gene. We now know that most genes that are involved in controlling cell death in C. elegans, have counterparts in humans.
Of importance for many research disciplines
The development of C. elegans as a novel experimental model system, the characterization of its invariant cell lineage, and the possibility to link this to genetic analysis have proven valuable for many research disciplines. For example, this is true for developmental biology and for analysis of the functions of various signaling pathways in a multicellular organism. The characterization of genes controlling programmed cell death in C. elegans soon made it possible to identify related genes with similar functions in humans. It is now clear that one of the signaling pathways in humans leading to cell death is evolutionarily well conserved. In this pathway ced-3-, ced-4- and ced-9-like molecules participate. Understanding perturbations in this and other signaling pathways controlling cell death are of prime importance for medicine.
Disease and programmed cell death
Knowledge of programmed cell death has helped us to understand the mechanisms by which some viruses and bacteria invade our cells. We also know that in AIDS, neurodegenerative diseases, stroke and myocardial infarction, cells are lost as a result of excessive cell death. Other diseases, like autoimmune conditions and cancer, are characterized by a reduction in cell death, leading to the survival of cells normally destined to die.
Research on programmed cell death is intense, including in the field of cancer. Many treatment strategies are based on stimulation of the cellular “suicide program”. This is, for the future, a most interesting and challenging task to further explore in order to reach a more refined manner to induce cell death in cancer cells.
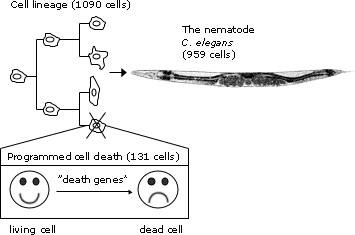 |
| Using the nematode C. elegans this year’s Nobel Laureates have demonstrated how organ development and programmed cell death are genetically regulated. They have identified key genes regulating programmed cell death and demonstrated that corresponding genes exist also in higher animals, including man. The figure schematically illustrates the cell lineage (top left) and the programmed cell death (below) in C. elegans. The fertilized egg cell undergoes a series of cell divisions leading to cell differentiation and cell specialization, eventually producing the adult organism (top right). In C. elegans, all cell divisions and differentiations are invariant, i.e. identical from individual to individual, which made it possible to construct a cell lineage for all cell divisions. During development, 1090 cells are generated, but precisely 131 of these cells are eliminated by programmed cell death. This results in an adult nematode (the hermaphrodite), composed of 959 somatic cells. |
Nobel Prizes and laureates
Six prizes were awarded for achievements that have conferred the greatest benefit to humankind. The 14 laureates' work and discoveries range from quantum tunnelling to promoting democratic rights.
See them all presented here.
