Press release

NOBELFÖRSAMLINGEN KAROLINSKA INSTITUTET
THE NOBEL ASSEMBLY AT THE KAROLINSKA INSTITUTE
The Nobel Assembly of Karolinska Institutet has today decided to award the Nobel Prize in Physiology or Medicine for 1984 jointly to
Niels K. Jerne, Georges J.F. Köhler and César Milstein
for theories concerning “the specificity in development and control of the immune system” and the discovery of “the principle for production of monoclonal antibodies”.
Summary
Niels K. Jerne is the great theoretician in immunology. In three main theories he has in a visionary way elucidated essential questions concerning specificity, development and regulation of the immune response. The natural-selection theory regarding antibody formation breaks with old views on the immune response and is a starting point of modern cellular immunology. His second theory explains how the cells of the immune system which mature in the thymus gland develop under the influence of the transplantation antigens of the host. The third, and most important theory, predicts how the immune response is regulated by a complicated network consisting of antibodies and anti-anti-bodies. The principles of the network theory are beginning to be exploited in prevention, diagnosis and treatment of disease.
The hybridoma technique for the production of monoclonal antibodies represents one of the most important methodological advances in biomedicine during the 1970s. An antibody producing cell and all its daughter cells produce an identical antibody molecule (monoclonal antibody). Since long scientists nourished the hope that it would become possible to produce monoclonal antibodies with predetermined specificities. This dream became a reality in 1975 when Georges J.F. Köhler and César Milstein described the hybridoma technique for production of monoclonal antibodies. They immortalized antibody producing cells by fusing them with tumour cells. The method allows unlimited production of monoclonal antibodies with predetermined specificity. Monoclonal antibodies has opened up completely new fields for theoretical and applied biomedical research and allows precise diagnosis and also treatment of disease.
The most important task for the immune system is to defend the body against bacteria, virus and other microorganisms. The specific defense is exerted by a subgroup of white blood cells (lymphocytes). The immune system needs to recognize and react specifically with a large number of foreign substances (antigens). How the lymphocytes develop these vital properties and how they build up the highly specialized recognition system of the immune apparatus has long been an area of intensive research.
Niels K. Jerne is the leading theoretician in immunology during the last 30 years. In three main theories he has elucidated central issues concerning specificity, development and regulation of the immune system in a comprehensive and convincing way. By his theories Jerne has outlined the development of modern immunology.
Theory 1: Specificity is predetermined
In his Natural-Selection Theory of Antibody Formation from 1955 Jerne explains the development of a specific antibody response in the following way. Each individual has a large number of natural antibodies with specificities for all antigens towards which the individual can respond. These antibodies develop already during fetal life in the absence of external antigens. The foreign antigen then selects the antibody molecule which has the best fit. The antigen-antibody binding stimulates the production of this particular antibody specificity.
Jerne’s natural-selection theory contrasted to the dogmatic views of the antibody response as formulated in the instruction theories which were prevailing at that time. According to these theories the antigen serves a template for the production of antibodies.
In Jerne’s natural selection theory it is implied that the generation of the enormous number of antibody specificities is independent of exogenous antigens. This view on the nature of the immune system constitutes the basis for modern immunology.
Theory 2: Reactivity against self-antigens creates diversity
The natural-selection theory is mainly concerned with the maturation of the immune system after it has acquired the ability to react with antigen. In the second theory on the Somatic Generation of Immune Recognition set forth in 1971 Jerne explains how the immune system develops from stem cells to mature lymphocytes which can react with antigen. He presupposes that every individual possesses all genes needed for the production of antibodies, and antibody-like molecules, which can bind all strong transplantation antigens of the species. Jerne suggests that lymphocytes mature in the thymus gland and in other lymphoid organs where they are exposed to the transplantation antigens of the individual. Cells which recognize the antigens are stimulated and enter cell division. As mutations accumulate in rapidly dividing cells new immunological specificities may develop. At the same time the specificities of the lymphocytes for self transplantation antigens are weakened. The mature lymphocytes will recognize foreign antigen associated with transplantation antigens. The theory explains how the immune system normally matures through the influence of self antigens. It also offers an explanation for the regulation of immunological specificity by genes belonging to the transplantation system.
Theory 3. Antibodies, anti-anti-bodies …
In his third main theory, the Network Theory from 1974, Jerne explains how the specific immune response is regulated. The theory has greatly stimulated research and led to new insights into the immune system. Recently its principles have been applied to diagnosis and treatment of disease.
A basis for the network theory was the observation that antibodies can elicit anti-antibodies directed against antigen binding structures on the first antibody (Figure 1). Moreover, anti-antibodies can stimulate the production of still another generation of antibodies, anti-anti-antibodies. Essentially, this antibody cascade is endless successively adding new specific properties to the immune system. The various antibody generations either stimulate or suppress the production of one another. Under normal conditions the network is balanced. When an antigen is introduced the equilibrium is disturbed. The immune system tries to restore balance which leads to an immune response against the antigen.
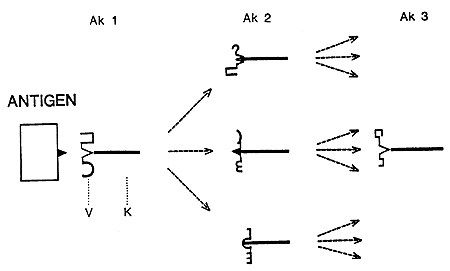 |
Figure 1. The Network Theory. Antibody 1 (Ak-1) has a structure in its variable (V) region which can bind the antigen. The V-region of Ak-1 contains unique structures which stimulate the production of various anti-antibodies (Ak-2). Some Ak-2 express V-region structures which mimic the antigen and which therefore can stimulate Ak-1 production.
Each antibody generation induces the production of still another and larger set of anti-antibodies in a cascade-like manner. The various sets of antibodies stimulate or suppress the production of each other in a complex network. Under normal conditions the network is balanced. However, the equilibrium is disturbed when an antigen is introduced and binds to Ak-1. The immune system attempts to restore the balance, i.e. it leads to an immune response.
Some examples where the network theory has been applied to experimental and clinical medicine are given in the following.
1. Infectious diseases. Anti-antibodies have been used in animals as a kind of vaccine against parasitic infections (trypanosomiasis), urinary tract infections, hepatitis and other infectious diseases.
2. Allergy. Anti-pollen antibodies may elicit allergic symptoms when an allergic person is exposed to pollen. The production of anti-pollen antibodies has been prevented in animals by anti-antibodies.
3. Autoimmune disease. Autoimmune disease may be caused by antibodies directed against the body’s own tissues. Experimental autoimmune disease has been successfully treated with anti-antibodies.
4. Transplantation. Anti-antiimmunity may be important in organ transplantation by contributing to immunological tolerance against antigen on the foreign graft.
5. Endocrinology. Anti-antibodies against hormones and hormone receptors may prevent binding of the hormone to the receptors. This has been described for insulin and its receptor.
6. Tumours. Anti-antibodies have been attempted as treatment of certain tumours of the human immune system.
Hybridoma – a technique for eternal production of monoclonal antibodies in cell cultures
Besides gene technology, which has already been honoured by several Nobel Prizes, the hybridoma technique represents the most important methodological advance within the field of biomedicine during the 1970s. The development of this technique is based on several observations concerning basic biological phenomena.
There are cells in the body – immune lymphocytes – which can produce millions of different antibodies. However, each single cell can only produce antibodies with a certain predetermined specificity. A prerequisite for the formation of a multitude of antibodies is, therefore, the existence of an excess of lymphocytes. If the body is exposed to a certain foreign antigen there may be stimulation of a lymphocyte which fortuitously has been endowed with the capacity to identify this particular antigen. This lymphocyte then starts to divide and forms a clone of cells which produces identical – monoclonal – antibodies.
The development of a clone of cells in connection with a normal immune response occurs under carefully controlled conditions. In rare cases, however, the body loses control over a clone of antibody producing cells. This may lead to formation of a special type of tumour (myeloma). Myeloma cells usually retain their capacity to produce a certain antibody, but because of the accidental emergence of the tumour one normally does not know with which antigen this antibody reacts.
White blood cells responsible for producing antibodies are highly specialized cells. As a consequence they lack capacity to survive for a longer time if they are removed from the body and incubated in a tissue culture medium. In contrast, myeloma cells can occasionally be cultivated continuously. Since long, biomedical research workers have nourished the dream to be able to propagate clones of cells which produce antibodies with predetermined specificity. This dream materialized when Georges J.F. Köhler and César Milstein in 1975 introduced the so-called hybridoma technology for production of monclonal antibodies. The principle features of the hybridoma technology is as follows (Figure 2).
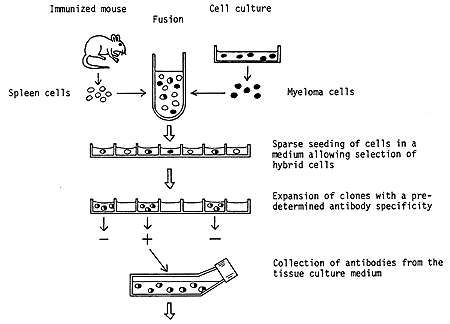
Figure 2. Principle steps in the production of a hybridoma. Spleen cells are prepared from animals, usually mice, which have been immunized with a selected antigen. These cells are then fused with myeloma cells maintained in culture in the laboratory. The product of this fusion is referred to as a hybridoma. Surprisingly, a hybrid of two cells can survive and also continue to divide. In this particular hybrid the myeloma cells contribute the capacity for survival, whereas the spleen cells direct the synthesis of antibodies with the preselected specificity. By special arrangements it is possible to achieve a multiplication of hybridoma cells but not of isolated myeloma cells. The hybrids obtained are propagated in a highly diluted state so that colonies deriving from single hybrid cells can be isolated. By use of a sensitive method the clones which produce the specific antibodies are identified. A particular hybridoma can then be used for future, unlimited production of a highly specific antibody.
The availability of monoclonal antibodies has opened completely new possibilities for basic as well as applied biomedical research. The following examples of the use of monoclonal antibodies can be given.
1. Detailed studies of the distribution of different functions in different parts of antigen molecules. These studies may concern building elements of infectious agents; cell products such as enzymes and hormones; surface structures of cells etc. The mapping of variations in the surface components of influenza virus which explain the occurrence of repeated infections is one example.
2. High degree purification of substances, e.g. interferon, by taking advantage of the unique capacity displayed by a particular monoclonal antibody to bind to a certain antigen. In this case one uses a technique referred to as affinity chromatography.
3. Diagnostic characterization of diseases by identification of special structures on the surface or on the inside of cells. Hereby it is possible to distinguish between different forms of tumours and follow the development of tumours. Furthermore, it is possible to distinguish between different kinds of normal white blood cells. This is of importance for the characterization of certain immune deficiency conditions as seen e.g. in connection with the disease AIDS (acquired immune deficiency syndrome).
Diseases caused by infectious agents can also be diagnosed by use of monoclonal antibodies. Thus, virus infected cells and bacteria or parasites inside or outside cells can be identified with a unique degree of specificity.
4. Treatment of diseases. Monoclonal antibodies against specialized white blood cells have been used with some success in connection with transplantation. There may also be possibilities to use monoclonal antibodies for treatment of tumours.
References
C. Milstein: Monoclonal Antibodies. Scientific American 1980, vol. 243, pp. 56-65.
Hybridomas: The Making of a Revolution. Science 1982, vol. 215, pp. 1073-1075.
Odödliga hybridceller – fabriker för tillverkning av monoklonala antikroppar. En av vår tids mest lovande medicinska upptäckter. Läkartidningen 1982, vol. 79, pp. 3545-3546.
L. Å. Hanson H. Wigzell: Immunologi, Del I. Teori. Almqvist & Wiksell, Stockholm, 1983.
M. Harboe & J. B. Natvig: Medisinsk Immunologi. Grøndahl & Søn Trykkeri A.s., Oslo, 1977.
Nobel Prizes and laureates
Six prizes were awarded for achievements that have conferred the greatest benefit to humankind. The 14 laureates' work and discoveries range from quantum tunnelling to promoting democratic rights.
See them all presented here.
