Press release

NOBELFÖRSAMLINGEN KAROLINSKA INSTITUTET
THE NOBEL ASSEMBLY AT THE KAROLINSKA INSTITUTE
9 October 1989
The Nobel Assembly at the Karolinska Institute has today decided to award the Nobel Prize in Physiology or Medicine for 1989 jointly to
J. Michael Bishop and Harold E. Varmus
for their discovery of “the cellular origin of retroviral oncogenes”.
Summary
The discovery awarded with this year’s Nobel Prize in Physiology or Medicine concerns the identification of a large family of genes which control the normal growth and division of cells. Disturbances in one or some of these so-called oncogenes (Gk ónco(s) bulk, mass) can lead to transformation of a normal cell into a tumor cell and result in cancer.
Michael Bishop and Harold Varmus used an oncogenic retrovirus to identify the growth-controlling oncogenes in normal cells. In 1976 they published the remarkable conclusion that the oncogene in the virus did not represent a true viral gene but instead was a normal cellular gene, which the virus had acquired during replication in the host cell and thereafter carried along.
Bishop’s and Varmus’ discovery of the cellular origin of retroviral oncogenes has had an extensive influence on the development of our knowledge about mechanisms for tumor development. Until now more than 40 different oncogenes have been demonstrated. The discovery has also widened our insight into the complicated signal systems which govern the normal growth of cells.
Cellular Oncogenes Discovered by the Use of Retrovirus
The term oncogene was introduced in the middle of the 1960s to denote special parts of the genetic material of certain viruses. It was believed that this part of the genetic material could direct the transformation of a normal cell into a tumor cell under the influence of other parts of the viral genetic material, alternatively via chemical or physical effects. The favourite theory of the time was that virus-mediated cell-to-cell transmittance of oncogenes was the origin of all forms of cancer. This view was later proven to be incorrect.
The original discovery of an oncogenic virus was made in 1916 by Peyton Rous working at the Rockefeller Institute in New York. Fifty years later Rous received the Nobel Prize in Physiology or Medicine. Rous virus, as the infectious agent later was named, is a member of a large virus family named retroviruses. The genetic material of these viruses is RNA (ribonucleic acid). This RNA can be transcribed into DNA (deoxyribonucleic acid) by a unique enzyme in the virus, reverse transcriptase. The 1975 Nobel Prize in Physiology or Medicine was awarded to David Baltimore, Renato Dulbecco and Howard Temin partly for the discovery of this enzyme.
Reverse transcription of the genetic material of the virus into DNA has the important consequence that it can become integrated into the chromosomal DNA in the cells. It was through investigations of Rous virus that this year’s laureates Michael Bishop and Harold Varmus in 1975 could demonstrate the true origin of oncogenes. They used one variant of Rous virus which contained an oncogenic gene (Figure 1) and another variant which lacked this gene. By use of these viruses they managed to construct a nucleic acid probe which selectively identified the oncogene. This probe was used to search for the corresponding genetic material in DNA from different cells. It was then found that oncogene-like material could be detected in different species throughout the animal kingdom, in fact even in simple organisms comprising only a few cells. Furthermore, it was shown that the gene had a fixed position in the chromosomes of a certain species, and that the gene, when it constituted part of the cellular genetic material, was divided into fragments (a mosaic gene) (Figure 1).
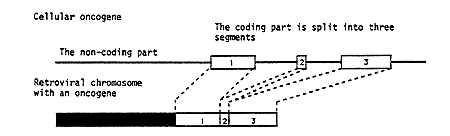 |
Figure 1. The difference between an oncogene in a virus and in a cell. In retroviruses causing tumors there is a separate segment of transforming nucleic acid which has been derived from a cell. The cellular gene is split (a mosaic gene) whereas the oncogene in the virus is continuous.
These findings led to the remarkable conclusion that the oncogene in the virus did not represent a true viral gene but a cellular gene which the virus had picked up far back during its replication in cells and carried along. This cellular gene was found to have a central function in the cells. It controlled their growth and division.
Through these studies of the abnormal, i.e. the diseased state, it was possible to elucidate critical normal cellular functions – a not uncommon situation in biomedical research. The original discovery of a cellular oncogene led to an intensive search for further similar genes. The explosive development of this field of research has led to the identification of more than 40 different oncogenes which direct different events in the complex signal systems that regulate the growth and division of cells. Changes in any one or more of these oncogenes may lead to cancer.
Balanced Cellular Interactions – A Biological Wonder
Symmetrical and asymmetrical, multicellular structures develop from the fertilized ovum by a process of differentiation about which only limited knowledge is available. In the fully developed individual carefully balanced conditions prevail. Damage of an organ elicits sophisticated repair processes which lead to restitution of the original condition of the organ. However, if a single cell escapes the network of growth control the result may be an abnormal local proliferation of cells or in the worst case a cancer implying the dissemination of cells running amok.
The development of a cancer is a complicated process involving several consecutive changes of the genetic material. Studies of cellular genes (proto-oncogenes) corresponding to the viral oncogenes, has started to shed light on the intricate systems which control normal cellular growth and division.
Cellular Oncogene Products Constitute Links in Signal Chains which Regulate Growth and Division of Cells
The regulation of growth and division of cells has turned out to be much more complicated than originally believed. Cellular oncogene products with different properties act in different positions of elaborate signal systems (Figure 2). In order to transmit signals from one cell to the other or from one cell to itself there are growth factors. These factors appear in the fluids surrounding cells. There are examples of oncogene products, viz. proteins produced in the cytoplasm, which can act as growth factors. Thus, it was found that the product of the sis1) gene was closely related to a previously identified growth factor PDGF (Platelet Derived Growth Factor).
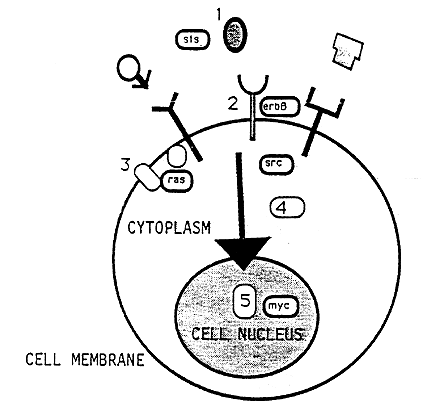
Figure 2. Oncogene products are links in signal chains that stretch from the cell surface to the genetic material in the cell nucleus. This chain is composed of (1) growth factors, (2) growth factor receptors, (3) signal transducing proteins in cell membranes, (4) phosphokinases in the cytoplasm and (5) proteins transported from the cytoplasm into the nucleus where they bind to DNA. The localization of different oncogene products (Sis, ErbB, Ras, Src, Myc) is schematically indicated.
In order for a growth factor to be able to interact with a cell there has to be membrane structures, receptors, to which they can bind. There are several oncogene products which represent receptors in the cytoplasmic membrane of the cells, e.g. ErbA, Fms, Kit. These receptors have a unique enzymatic activity. They are so-called kinases with a capacity to phosphorylate (=add a phosphate group) the amino acid tyrosine. There are two more groups of oncogene products with phosphokinase activity; firstly tyrosine/phosphokinase which lack receptor function and is located at the inside of the cytoplasmic membrane, and secondly serine/threonine phosphokinase which is found in the cytoplasm.
Thus, oncogene products function as links in signal chains stretching from the surface of the cell to the genetic material in the nucleus. In the cytoplasm there is one more group of oncogene products. These are called Ras and are related to important cellular signal factors called G-proteins.
Finally, there is a large number of oncogene products which are located in the nucleus of the cell, i.e. Myc, Myb, Fos, ErbA and others. These products direct the transcription of DNA into RNA and therefore play a critical role in the selection of proteins to be synthesized by the cell.
Cancer – A Complex, Biological Sequence of Events
Changes in the genetic material constitute the basis for the development of all cancer. Generally there are several consecutive such changes which influence different steps in the signal chains described above. Therefore, one should à priori not expect to find one single clue to the mechanism of origin of cancer. However, application of the expanding knowledge in the oncogene field allows us to start comprehending the disharmonic orchestration behind abnormal cellular growth.
It is conceptually incorrect to speak about “cancer genes”. However, historical circumstances explain why the oncogene terminology was introduced before a designation of the corresponding normal cellular genes was proposed. From the point of view of cancer the important matter is to compare oncogenes in normal cells and in tumor cells.
Oncogenes as a Cause of Cancer
The majority of oncogenes have been discovered in experimental studies using retroviruses. However, in a few cases oncogenes were identified by the use of an alternative technique, i.e. genetic material was isolated from tumor cells of non-viral origin and transferred (transfected) to other cells prapagated in culture. The cells receiving the DNA changed growth pattern, and further characterization of the transfected genetic material revealed the presence of oncogenes.
Two principally different forms of activation of oncogenes can be distinguished. Firstly, the normal cellular oncogene is hyperactive, and secondly the oncogene product is altered so that it can no longer be regulated in a normal way. There are several examples of these types of activation of oncogenes.
The discovery of oncogenes was as mentioned originally made by the use of retroviruses. This infers that genetic control elements in the virus itself can be responsible for the abnormal expression of the oncogene. However, in many cases it was found that alterations of the transferred oncogene contributed to its accentuated expression.
There are retroviruses which lack oncogenes but still can induce cancer. This is due to the fact that the virus has inserted its genetic material (in the form of DNA) very close to a normally occurring oncogene in the genetic material of the cell. This may result in an increased turn-over of the oncogene which may lead to abnormal cellular growth. The corresponding phenomenon can also occur in the absence of retroviruses. In this case there is a reorganization of the genetic material in the cell. Such a reorganization may occur within a single chromosome or by exchange of material between chromosomes. Repeated copying of a normal oncogene can lead to its amplification in the chromosome and consequently to increased amounts of the oncogene product. In certain brain tumors, glioblastomas, an amplified erbB-gene has been found, and a correspondingly increased neu-gene activity was shown in some forms of breast cancer.
The same effect can be seen when there is a reciprocal exchange of segments between chromosomes (translocation). Thus the normal myc-gene on chromosome 8 has been translocated to chromosome 14 in many patients with Burkitt’s lymphomas (Figure 3). The insertion of the myc-gene containing chromosome segment is such that it becomes located close to hyperactive genes directing the synthesis of antibody protein. As a consequence the myc-gene becomes activated. Chromosome translocations occur in many different tumors. Chromosome analysis can therefore be of considerable value for localization of genetic changes in the genome critical for tumor development.
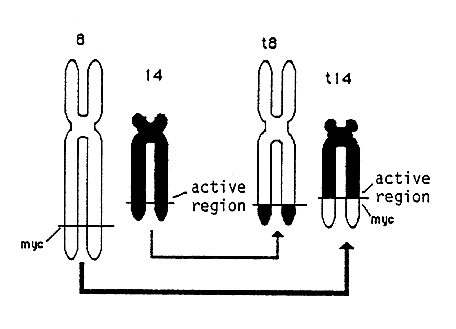 |
Figure 3. Chromosome translocation in Burkitt’s lymphoma. Segments have been exchanged between chromosomes 8 and 14 which has activated the oncogene myc.
Oncogenes with point mutations have been observed in many tumors. These mutations may cause alterations in the amino acid composition of the gene product. A well-known example of such a modification is the exchange of amino acid 12 from glycine to valine in the ras gene product which has been observed in human tumor material. The mutation may also be somewhat more extensive leading to the absence of part of the protein (deletion). Different examples of modified oncogenes in human tumor material are given in Table I.

The Importance of Viruses for Cancer in Man
Cancer is not a contagious disease. However, infectious agents like viruses can contribute to the origin of cancer. Thus, it is by use of retroviruses that most oncogenes were identified, the starting materials in such investigations often being highly specialized, experimentally derived tumors. It seems likely that retroviruses play a relatively limited role for the development of cancer under natural conditions. The only known example in man in which a retrovirus infection contributes to the origin of cancer is the HTLV-1 associated lymphomas which occur in Japan.
However, there are other kinds of viruses which can contribute to the development of tumors in man. All these viruses have DNA as their genetic material. As examples can be mentioned papillome (wart) viruses and Epstein-Barr virus, a type of herpes virus. Certain types of papillome viruses play a role for the development of cervical cancer in the genital tract, while Epstein-Barr virus is an important factor for the development of Burkitt’s lymphomas in Africa and nasopharyngeal cancer in Asia. However, in all these cases factors in addition to the virus infections are required for the cancer to develop.
References
J.M. Bishop: Oncogenes. Scientific American, 1982, 246, 68-78.
T. Hunter: The Proteins of Oncogenes. Scientific American, 1984, 251, 60-69.
C-H. Heldin & B. Westermark: Tillväxtfaktorer och onkgener. Läkartidningen 1988, 85, 497-499.
E. Norrby: I: Våra virus. Virus och cancer. Allmänna Förlaget, 1987, sid. 66-74.
1) All oncogenes are identified by the use of three letter abbreviations. In addition cellular and viral oncogenes are sometimes distinguished by c- and v- prefixes, respectively, e.g. c-src and v-src.
Nobel Prizes and laureates
Six prizes were awarded for achievements that have conferred the greatest benefit to humankind. The 14 laureates' work and discoveries range from quantum tunnelling to promoting democratic rights.
See them all presented here.
