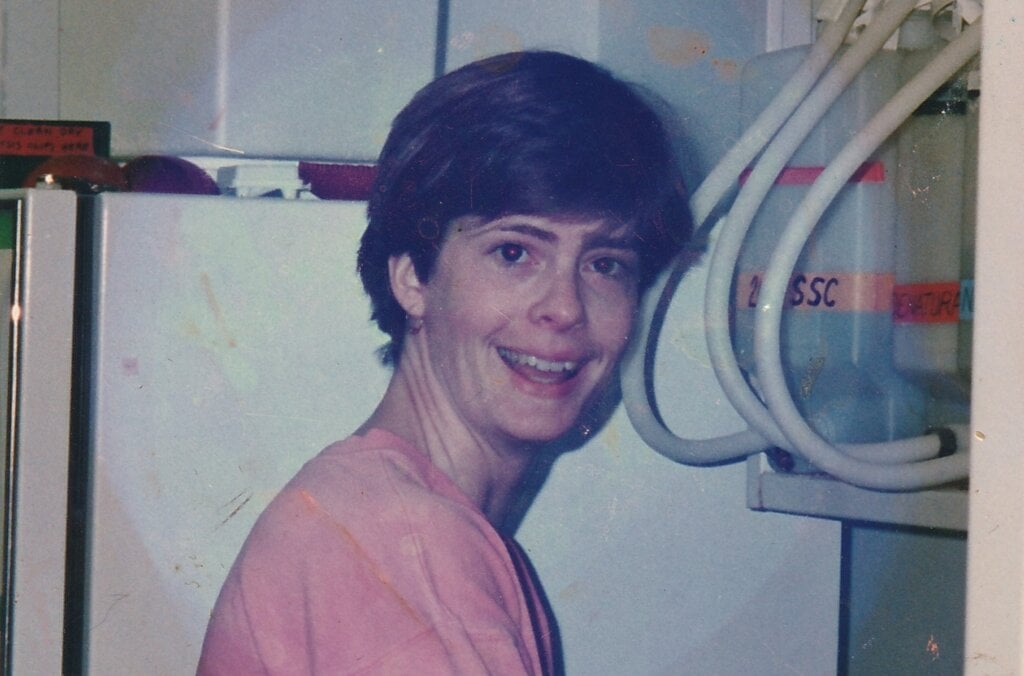
MARY BRUNKOW
Nobel Prize in Physiology or Medicine 2025
A chance discovery made around 80 years ago paved the way for Mary Brunkow’s painstaking work, which contributed to redefining how the immune system functions. From investigating an obscure gene dismissed as ‘junk’ by other scientists, to choosing to opt out of academia and conduct her research at a biotech startup, Brunkow has embraced the unexpected throughout her career.



Mary Brunkow was born in 1961 in Portland, Oregon, the middle child of three. In high school, she enjoyed basketball and ran for the track and cross-country teams, but was, by her own omission, pretty bad at both. She was, however, a good student with a love of maths and science, especially genetics.
Brunkow longed to go to university in a big city and, after graduation, started on the pre-med track at the University of Washington. She was inspired by her paternal grandfather who was a doctor and surgeon, and had a strong desire to help people. However, after taking a genetics class in her final year at university and getting her first taste of lab work, she realised research was where her future lay. “The minute I walked into that research laboratory, it was just an environment that really struck me,” she said.
“I like the idea of something really concrete and coming up with a discovery that proves something.”
Mary Brunkow
The lively and collaborative atmosphere of the lab stirred Brunkow to continue her studies at Princeton University as one of the first graduate students to join its department of molecular biology. She made the brave decision to focus on a seemingly obscure gene dismissed as ‘junk’ by other scientists for her PhD, before carrying out postdoctoral research at the University of Toronto.

While her fellow postdocs stayed in academia, Brunkow wanted to do something with a more direct application to human health. It was an exciting time in science. The Human Genome Project – an ambitious international research effort aimed at deciphering the chemical makeup of the entire human genetic code – was running at full speed and new genomics-related technologies were taking off. Her postdoctoral research over, she decided to take another unexpected path, joining a young biotechnology company in a Seattle suburb that developed pharmaceuticals for autoimmune diseases. According to Brunkow, the competition and self-promotion she had seen in academia was absent. “Everybody was working towards the same goal of finding new drug targets that would eventually be hopefully developed into drugs,” she said. There, Brunkow learned of a mouse genetics programme set up following the Second World War to understand how the effects of radiation might impact human health and alter genes.
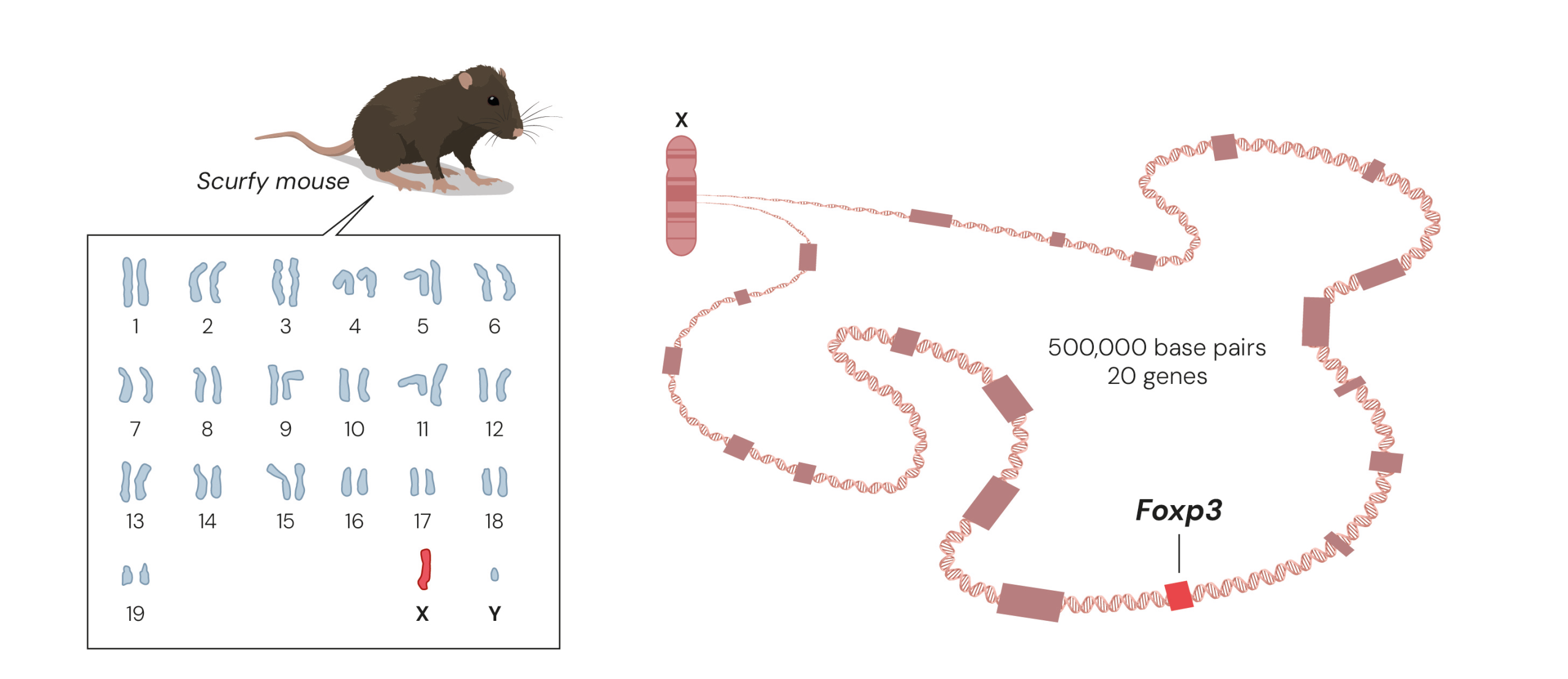
A chance discovery of a spontaneous genetic mutation in mice made almost 80 years ago turned out to be the foundation for Brunkow and her colleague Fred Ramsdell’s Nobel Prize-worthy work. The mutation, known as scurfy, only affects male mice who show several hallmarks of severe autoimmune disease, where the immune system attacks almost every organ in their body. Yet the gene that caused the serious symptoms was unknown. The duo realised that if they could understand the molecular mechanism underlying the mice’s disease, they could gain decisive insights into how autoimmune diseases arise.
Today, it is possible to map a mouse’s entire genome and find a mutated gene in a few days, but in the 1990s it was like looking for a needle in a huge haystack. Mapping had shown that the scurfy mutation must be somewhere in the middle of the X chromosome, so Brunkow and Ramsdell painstakingly mapped that area of the X chromosome, eventually narrowing their search and identifying 20 possible target genes. “It was really a molecular slog to get to that exact mutation, because it was just a very small genetic alteration that results in quite a profound change in the immune system,” Brunkow said.
She bred her own colony of scurfy mice from the same line, taking over and adapting a disused janitor’s closet to provide space to do so. By comparing the genes in healthy mice and scurfy mice they discovered a minor change in a gene that would later be named Foxp3.
During their work, Brunkow and Ramsdell had begun to suspect that a rare autoimmune disease, IPEX, which is also linked to the X chromosome, might be the human variant of the scurfy mice’s disease. In 2001, they revealed that mutations in the Foxp3 gene cause both the human disease called IPEX and the scurfy mice’s ill health.
In 2025, Brunkow and Ramsdell shared the Nobel Prize in Physiology or Medicine with Japanese immunologist Shimon Sakaguchi. Their discoveries launched the field of peripheral tolerance, spurring the development of medical treatments for cancer and autoimmune diseases.
Embracing the unexpected and following the path less travelled has yielded Brunkow a fascinating and inspiring career, combining academic research, biotech startups, consulting, science communication and programme management.
“Discoveries come from places where you never imagined, so you have to keep an open mind and keep your eyes open to different pathways, as you’re journeying through a topic of interest, but also an open mind in terms of career path and where you go in life.”
Mary Brunkow
“I would say I’m a pretty good example of … one who has kind of changed directions and not stayed on one single track. Sometimes doors open off to the side a little bit. There’s nothing wrong with exploring different options.”


Women who changed science
Nobel Prizes and laureates
Six prizes were awarded for achievements that have conferred the greatest benefit to humankind. The 14 laureates' work and discoveries range from quantum tunnelling to promoting democratic rights.
See them all presented here.
