Press release
English
English (pdf)
Swedish
Swedish (pdf)
![]()
6 October 2008
The Nobel Assembly at Karolinska Institutet has today decided to award
The Nobel Prize in Physiology or Medicine for 2008 with one half to
Harald zur Hausen
for his discovery of “human papilloma viruses causing cervical cancer”
and the other half jointly to
Françoise Barré-Sinoussi and Luc Montagnier
for their discovery of “human immunodeficiency virus”
Summary
This year’s Nobel Prize awards discoveries of two viruses causing severe human diseases.
Harald zur Hausen went against current dogma and postulated that oncogenic human papilloma virus (HPV) caused cervical cancer, the second most common cancer among women. He realized that HPV-DNA could exist in a non-productive state in the tumours, and should be detectable by specific searches for viral DNA. He found HPV to be a heterogeneous family of viruses. Only some HPV types cause cancer. His discovery has led to characterization of the natural history of HPV infection, an understanding of mechanisms of HPV-induced carcinogenesis and the development of prophylactic vaccines against HPV acquisition.
Françoise Barré-Sinoussi and Luc Montagnier discovered human immunodeficiency virus (HIV). Virus production was identified in lymphocytes from patients with enlarged lymph nodes in early stages of acquired immunodeficiency, and in blood from patients with late stage disease. They characterized this retrovirus as the first known human lentivirus based on its morphological, biochemical and immunological properties. HIV impaired the immune system because of massive virus replication and cell damage to lymphocytes. The discovery was one prerequisite for the current understanding of the biology of the disease and its antiretroviral treatment.
Discovery of human papilloma virus causing cervical cancer
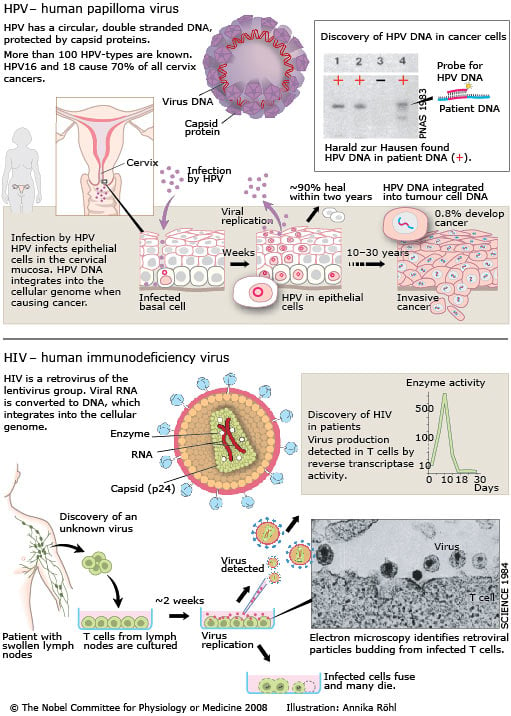
Against the prevailing view during the 1970s, Harald zur Hausen postulated a role for human papilloma virus (HPV) in cervical cancer. He assumed that the tumour cells, if they contained an oncogenic virus, should harbour viral DNA integrated into their genomes. The HPV genes promoting cell proliferation should therefore be detectable by specifically searching tumour cells for such viral DNA. Harald zur Hausen pursued this idea for over 10 years by searching for different HPV types, a search made difficult by the fact that only parts of the viral DNA were integrated into the host genome. He found novel HPV-DNA in cervix cancer biopsies, and thus discovered the new, tumourigenic HPV16 type in 1983. In 1984, he cloned HPV16 and 18 from patients with cervical cancer. The HPV types 16 and 18 were consistently found in about 70% of cervical cancer biopsies throughout the world.
Importance of the HPV discovery
The global public health burden attributable to human papilloma viruses is considerable. More than 5% of all cancers worldwide are caused by persistent infection with this virus. Infection by the human papilloma virus is the most common sexually transmitted agent, afflicting 50-80% of the population. Of the more than 100 HPV types known, about 40 infect the genital tract, and 15 of these put women at high risk for cervical cancer. In addition, HPV is found in some vulval, penile, oral and other cancers. Human papilloma virus can be detected in 99.7% of women with histologically confirmed cervical cancer, affecting some 500,000 women per year.
Harald zur Hausen demonstrated novel properties of HPV that have led to an understanding of mechanisms for papilloma virus-induced carcinogenesis and the predisposing factors for viral persistence and cellular transformation. He made HPV16 and 18 available to the scientific community. Vaccines were ultimately developed that provide ≥95 % protection from infection by the high risk HPV16 and 18 types. The vaccines may also reduce the need for surgery and the global burden of cervical cancer.
Discovery of HIV
Following medical reports of a novel immunodeficiency syndrome in 1981, the search for a causative agent was on. Françoise Barré-Sinoussi and Luc Montagnier isolated and cultured lymph node cells from patients that had swollen lymph nodes characteristic of the early stage of acquired immune deficiency. They detected activity of the retroviral enzyme reverse transcriptase, a direct sign of retrovirus replication. They also found retroviral particles budding from the infected cells. Isolated virus infected and killed lymphocytes from both diseased and healthy donors, and reacted with antibodies from infected patients. In contrast to previously characterized human oncogenic retroviruses, the novel retrovirus they had discovered, now known as human immunodeficiency virus (HIV), did not induce uncontrolled cell growth. Instead, the virus required cell activation for replication and mediated cell fusion of T lymphocytes. This partly explained how HIV impairs the immune system since the T cells are essential for immune defence. By 1984, Barré-Sinoussi and Montagnier had obtained several isolates of the novel human retrovirus, which they identified as a lentivirus, from sexually infected individuals, haemophiliacs, mother to infant transmissions and transfused patients. The significance of their achievements should be viewed in the context of a global ubiquitous epidemic affecting close to 1% of the population.
Importance of the HIV discovery
Soon after the discovery of the virus, several groups contributed to the definitive demonstration of HIV as the cause of acquired human immunodeficiency syndrome (AIDS). Barré-Sinoussi and Montagnier’s discovery made rapid cloning of the HIV-1 genome possible. This has allowed identification of important details in its replication cycle and how the virus interacts with its host. Furthermore, it led to development of methods to diagnose infected patients and to screen blood products, which has limited the spread of the pandemic. The unprecedented development of several classes of new antiviral drugs is also a result of knowledge of the details of the viral replication cycle. The combination of prevention and treatment has substantially decreased spread of the disease and dramatically increased life expectancy among treated patients. The cloning of HIV enabled studies of its origin and evolution. The virus was probably passed to humans from chimpanzees in West Africa early in the 20th century, but it is still unclear why the epidemic spread so dramatically from 1970 and onwards.
Identification of virus−host interactions has provided information on how HIV evades the host’s immune system by impairing lymphocyte function, by constantly changing and by hiding its genome in the host lymphocyte DNA, making its eradication in the infected host difficult even after long-term antiviral treatment. Extensive knowledge about these unique viral host interactions has, however, generated results that can provide ideas for future vaccine development as well as for therapeutic approaches targeting viral latency.
HIV has generated a novel pandemic. Never before has science and medicine been so quick to discover, identify the origin and provide treatment for a new disease entity. Successful anti-retroviral therapy results in life expectancies for persons with HIV infection now reaching levels similar to those of uninfected people.
Harald zur Hausen, born 1936 in Germany, German citizen, MD at University of Düsseldorf, Germany. Professor emeritus and former Chairman and Scientific Director, German Cancer Research Centre, Heidelberg, Germany.
Françoise Barré-Sinoussi, born 1947 in France, French citizen, PhD in virology, Institut Pasteur, Garches, France. Professor and Director, Regulation of Retroviral Infections Unit, Virology Department, Institut Pasteur, Paris, France.
Luc Montagnier, born 1932 in France, French citizen, PhD in virology, University of Paris, Paris, France. Professor emeritus and Director, World Foundation for AIDS Research and Prevention, Paris, France.
Nobel Prizes and laureates
Six prizes were awarded for achievements that have conferred the greatest benefit to humankind. The 14 laureates' work and discoveries range from quantum tunnelling to promoting democratic rights.
See them all presented here.
