The structure of a photosynthetic reaction center
In 1984, after having analyzed the X-ray diffraction pattern from the reaction center crystals, Johann Deisenhofer, Robert Huber and Hartmut Michel could present the 3-dimensional structure of the reaction center, the first high-resolution structure of a membrane protein and also the most complex molecular structure which had been solved.
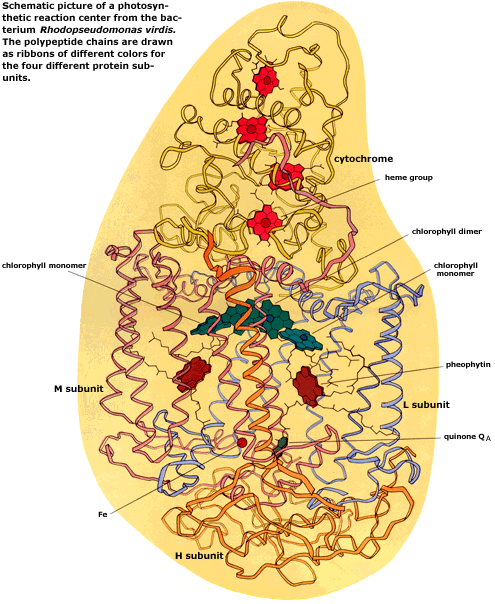
The reaction center is composed of four protein subunits. Two of these, the L and M subunits, each form five membrane-spanning helices. The structure shows the precise arrangement in the L and M subunits of the photochemically active groups – two chlorophyll molecules forming a dimer, two monomeric chlorophylls, two pheophytin molecules (these lack the central magnesium ion of chlorophyll), one quinone molecule, called QA (a second quinone molecule, QB, is lost during the preparation of the reaction center) and one iron ion (Fe). The L and M subunits and their chromophores are related by a twofold symmetry axis that passes through the chlorophyll dimer and the iron.
A third subunit, H, without active groups and located on the membrane inner surface, is anchored to the membrane by a protein helix.
The remaining subunit, a cytochrome with four heme groups (related to the blood pigment hemoglobin), binds at the outer surface of the membrane.
 |
Nobel Prizes and laureates
Six prizes were awarded for achievements that have conferred the greatest benefit to humankind. The 14 laureates' work and discoveries range from quantum tunnelling to promoting democratic rights.
See them all presented here.
