Press release

17 October 1990
The Royal Swedish Academy of Sciences has decided to award the 1990 Nobel Prize in Chemistry to
Professor Elias James Corey, Harvard University, Cambridge, Massachusetts,
USA,
for his development of the theory and methodology of organic synthesis.
Prize for masterly development of organic synthesis
The development of the art of organic synthesis during little over a hundred years has afforded efficient methods of manufacturing products such as plastics and other artificial fibres, paints and dyes, biocides and pharmaceutical products, all of which have contributed to the high standards of living and health, and the longevity, enjoyed at least in the Western world.
This year’s Nobel Prize in Chemistry has been awarded to Professor Elias J. Corey, USA, for his important contributions to synthetic organic chemistry. He has developed theories and methods that have made it possible to produce a large variety of biologically highly active, complicated natural products, thereby making, among other things, certain pharmaceuticals commercially available. Corey’s work has also led to new general methods of producing, synthesising, compounds in simpler ways.
The background to Elias J. Corey’s successes lies in the fact that he has in a strictly logical way developed the principles of what is termed retrosynthetic analysis. This involves starting from the planned structure of the molecule one wishes to produce, the target molecule, and analysing what bonds must be broken, thus simplifying the structure step by step. One then finds that certain fragments are already known and their structure and synthesis already described. After working backwards in this way from the complex to the already known, it is possible to start building, synthesising, the molecule. This method has proved very amenable to data processing, which has entailed rapid developments in synthesis planning. Combining this synthesis planning with singular creativity, Corey has developed new methods of synthesis. He has produced some hundred important natural products, for example the active substance in an extract from the ginkgo tree, used in folk medicine in China.
Background information
Organic synthesis, that is, the production of complicated organic compounds using simple and cheap starting material, is one of the prerequisites of our civilisation. It is understandable that contributions in this field have often been rewarded with the Nobel Prize in Chemistry. Thus in 1902, only the second year that Nobel Prizes were awarded, the Chemistry Prize went to Emil Fischer for his work on synthesis within sugar and purine chemistry. In 1905 Adolf von Baeyer received the prize in recognition of contributions to the development of the chemical industry through his work on organic dyestuffs. Otto Wallach received the 1910 Prize for contributions to the development of the chemical industry. The 1912 prize went to Victor Grignard for his development of organic magnesium compounds, also termed Grignard reagents, into important intermediates in organic synthesis. In 1950 Otto Diels and Kurt Alder shared the Nobel Prize for discovering the preparatively very useful diene synthesis. Robert B. Woodward received the 1965 prize for his brilliant contributions to the development of the art of organic synthesis. In 1979 Herbert C. Brown and Georg Wittig were rewarded for developing boron compounds and phosphorus compounds, respectively, into important reagents in organic synthesis.
The synthesis of complicated organic compounds often shows elements of artistic creation, as for example architecture. Many earlier syntheses were performed more or less intuitively, so that their planning was difficult to perceive. Asking a chemist how he came upon precisely the starting materials and reactions that so elegantly led to the desired result would probably be as meaningless as asking Picasso why he painted as he did. The process of synthetic planning has been likened to a game of three-dimensional chess using 40 pieces on each side. But the problem may be even harder than this. Over 35,000 usable methods of synthesis are described in chemical literature, each with its possibilities and its limitations. During the synthesis, moreover, new methods appear which can modify the strategy.
Beginning in the 1960’s, Corey coined the term, and developed the concept of, retrosynthetic analysis. Starting from the structure of the molecule he was to produce, the target molecule, he established rules for how it should be dissected into smaller parts, and what strategic bonds should be broken. In this way, less complicated building blocks were obtained, which could later be assembled in the process of synthesis. These building blocks were then analysed in the same way until simple compounds had been reached, whose synthesis was already described in the literature, or which are commercially available. Corey showed that strictly logical retrosynthetic analysis was amenable to computer programming. At present, synthesis planning with the help of computers is developing rapidly.
Through his brilliant analysis of the theory of organic synthesis, Corey has contributed in high degree to his own and other researchers’ being able, during the last few decades, to complete total syntheses, hitherto impossible, of complicated, naturally-occurring, biologically active compounds, according to simple logical principles.
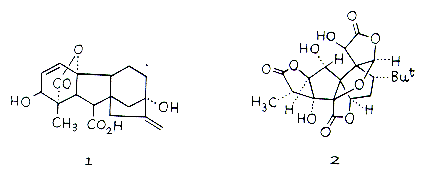
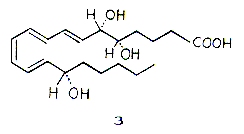
Elias J. Corey has himself synthesised about a hundred important natural products, of which only a few will be mentioned here. In 1978 he produced gibberellic acid (1), which belongs to a class of very important plant hormones of complicated structure. Recently, he has also synthesised (+)-ginkgolid (2), which owing to its complicated structure is a formidable challenge to anyone working in synthetic chemistry. (+)-ginkgolid is the active substance in an extract from the ginkgo tree, used as a folk medicine in China. The sales value of this natural product is believed to amount to $500 million annually. It is used in treatment of blood circulation disturbances in the elderly, and in asthma. Corey’s most important total syntheses concern the medically very important eicosanoids such as prostaglandins, prostacyclins, thromboxanes and leucotrienes, which occur naturally in extremely small quantities. These frequently very unstable compounds answer for multifarious and vital regulatory functions of significance for reproduction, blood coagulation, normal and pathological processes in the immune system, etc. Their importance is witnessed by the award of the 1982 Nobel Prize in Physiology or Medicine to Sune Bergstrom, Bengt Samuelsson and Sir John Vane for the discovery of prostaglandins and closely related biologically active substances. Corey has with enormous skill carried out structural determination and total syntheses of a large number of compounds of many different types of eicosanoids such as prostaglandins and leucotrienes such as lipoxin A (3). It is thanks to Corey’s contributions that many of these important pharmaceuticals are commercially available.
 |
To perform the total syntheses successfully, Corey was also obliged to develop some fifty entirely new or considerably improved synthesis reactions or reagents. It is probable that no other chemist has developed such a comprehensive and varied assortment of methods which, often showing the simplicity of genius, have become commonplace in the synthesising laboratory. His systematic use of different types of organometallic reagent has revolutionised recent techniques of synthesis in many respects. He has also in recent years introduced a number of very effective enzyme-like catalysts. These chiral catalysts give only one mirror isomer of the target product, in certain types of synthetically important reaction. The chiral catalysts are simple and easy to recover, and can in some cases be used in their own production.
Nobel Prizes and laureates
Six prizes were awarded for achievements that have conferred the greatest benefit to humankind. The 14 laureates' work and discoveries range from quantum tunnelling to promoting democratic rights.
See them all presented here.
