Press release

16 October 1991
The Royal Swedish Academy of Sciences has decided to award the 1991 Nobel Prize in Chemistry
to Professor Richard R. Ernst, Eidgenössische Technische Hochschule (ETH), Zürich, Switzerland,
for his contributions to the development of the methodology of high resolution nuclear magnetic resonance (NMR) spectroscopy.
Revolutionary developments make a spectroscopic technique indispensable for chemistry
The 1991 Nobel Prize in Chemistry has been awarded to Professor Richard R. Ernst of the ETH, Zurich, for important methodological developments within nuclear magnetic resonance (NMR) spectroscopy. NMR spectroscopy has during the last twenty years developed into perhaps the most important instrumental measuring technique within chemistry. This has occurred because of a dramatic increase in both the sensitivity and the resolution of the instruments, two areas in which Ernst has contributed more than anybody else.
NMR spectroscopy is today used within practically all branches of chemistry, at universities as well as industrial laboratories. The method has its most important applications as a tool for the determination of molecular structure in solution. It can today be applied to a wide variety of chemical systems, from small molecules (e.g. drugs) to proteins and nucleic acids. Further, chemists use NMR to study interactions between different molecules (e.g. enzyme – substrate, soap – water), to investigate molecular motion, to get information on the rate of chemical reactions and for many other problems. The NMR technique is today also important in related sciences, such as physics, biology and medicine.
Background
The first successful NMR experiments were reported in 1945, by two independent groups in the USA (Bloch and co-workers at Stanford and Purcell with his group at Harvard). Their discovery was awarded a Nobel Prize in Physics in 1952. The NMR phenomenon can be explained in the following way. When matter is placed in a magnetic field, some of the atomic nuclei (e.g. nuclei of hydrogen atoms, called protons) behave like microscopic compass needles. These tiny compass needles (called nuclear spins) can, according to the laws of quantum mechanics, orient themselves with respect to the magnetic field in only a few ways. These orientations are characterized by different energy levels. The nuclear spins can be forced to jump between levels if the sample is exposed to radio waves of exactly specified frequency. The frequency is varied during the course of the experiment and, when it exactly matches the characteristic frequency of the nuclei (the resonance frequency), an electric signal is induced in the detector. The strength of the signal is plotted as a function of frequency in a diagram called the NMR spectrum. Around 1950, it was discovered that nuclear resonance frequencies depended not only on the nature of the atomic nuclei, but also on their chemical environment. The possibility of using NMR as a tool for chemical analysis soon became obvious and was mentioned by, among others, Professor Purcell in his 1952 Nobel lecture. A fundamental difficulty in the early days was the relatively low sensitivity of the NMR method.
A major breakthrough occurred in 1966 when Ernst (together with Weston A. Anderson, USA) discovered that the sensitivity of NMR spectra could be increased dramatically if the slow radiofrequency sweep that the sample was exposed to was replaced by short and intense radiofrequency pulses. The signal was then measured as a function of time after the pulse. The next pulse and signal acquisition were started after a few seconds, and the signals after each pulse were summed in a computer. The NMR signal measured as a function of time is not amenable to a simple interpretation (see Figure la). It is however possible to analyze what resonance frequencies are present in such a signal – and to convert it to an NMR spectrum – by a mathematical operation (Fourier transformation, FT) performed rapidly in the computer. The result of the Fourier transformation of Figure la is shown in Figure lb.
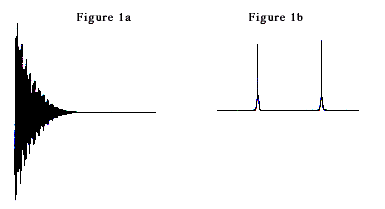
This discovery is the basis of modern NMR spectroscopy. The ten-fold, and sometimes hundred-fold, increase in sensitivity has made it possible to study small amounts of material as well as chemically interesting isotopes of low natural occurrence, e.g. carbon- 13. The enormous potential of the new technique – called FT NMR – quickly became obvious to NMR spectroscopists. The chemical research community got access to it in the early seventies through commercial FT NMR instruments. Nowadays, practically no other types of NMR spectrometer are manufactured.
By the end of the sixties, NMR spectroscopists had begun to use new magnet designs, based on superconducting materials, and the quality of spectra – expressed both in terms of sensitivity and resolution – improved quickly during the seventies. Consequently, more complex systems could be studied and more sophishcated questions answered. To move to very large molecules, macromolecules, another breakthrough was necessary, and this again carried the signature of Ernst. Inspired by a lecture of Jean Jeener, Belgium, at a summer school at the beginning of the seventies, Ernst and co-workers showed in 1975-76 how “two-dimensional” (2D) NMR experiments could be performed and demonstrated that 2D FT NMR opened entirely new possibilities for chemical research.
This 2D methods functions in the following way. Nuclear spins in a magnetic field are now subjected to sequences of radio-frequency pulses rather than to single pulses. The time course of the experiment is divided into four intervals. During the “preparation period”, the equilibrium of the nuclear spin system is distorted by one or several pulses. This non-equilibrium is allowed to evolve for a certain time (the “evolution period”), after which the next series of pulses (the “mixing period”) leads to the “detection period”. Here the NMR signal is detected as a function of time in the same way as in ordinary, one-dimensional FT NMR. After this, one moves to the next preparation period and repeats the experiment with different evolution period. The change in the evolution period causes the signal measured during the detection period to change. One might say that the history of spins during the evolution period becomes encoded in the variation of the signal measured during the detection period. This gives a two-dimensional table with signal intensity as a function of both the point in time during the detection period and the length of the evolution period. Finally, the Fourier transformation is performed twice – with respect to both these time parameters – to obtain a two-dimensional frequency spectrum in the form of a map of the dependence of the signal intensity on two frequency parameters (denoted f1 and f2 in Figure 2).
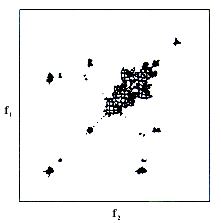 |
| Figure 2. A so-called 2D correlation spectrum for protons in cane sugar shows which signals originate from nuclei that are close to each other. |
Introduction of the second frequency dimension allows the spectral information to attain much higher resolution – like looking at the skyline of a mountain range and then looking at the whole range from an aircraft above. Depending on the design of the preparation and the mixing periods, one obtains a variety of 2D NMR experiments. Some are used to spread the information over two dimensions rather than one (separation of interactions) while others are designed to find which nuclei have some form of contact with each other (correlation of signals).
In the mid-seventies, Ernst also proposed a method of obtaining NMR-tomographic images which became one of the most common (the NMR tomography method as such was earlier realized by Lauterbur in the USA, Mansfield in England and others).
Since the mid-seventies, Ernst and co-workers have continuously and decisively contributed to the development of NMR spectroscopy, and in particular its two-, and more recently three- and multi-dimensional varieties. Applications of his methods were soon to come. For example, it has become possible over the past ten years to use NMR to determine the three-dimensional structure of organic and inorganic compounds as well as proteins and other biological macromolecules in solution with an accuracy comparable to what can be attained in crystals using X-ray diffraction. Interactions between biological molecules and other substances (metal ions, water, drugs) have also been studied in detail. Other important chemical applications are identification of chemical species (where NMR spectra act as the fingerprint of a molecule), studies of rates of certain chemical reactions and of molecular motions in the liquid state. In the border area between chemistry and biology, NMR is being used to study how metabolic processes are influenced by drugs, ischaemia etc. Ernst’s own work often falls in the border area between chemistry and physics and can, if one so wishes, be treated as extremely elegant experimental verification of the correctness of quantum mechanics.
Nobel Prizes and laureates
Six prizes were awarded for achievements that have conferred the greatest benefit to humankind. The 14 laureates' work and discoveries range from quantum tunnelling to promoting democratic rights.
See them all presented here.
