Press release
English
Swedish
German

11 October 1995
The Royal Swedish Academy of Sciences has decided to award the 1995 Nobel Prize in Chemistry to
Professor Paul Crutzen, Max-Planck-Institute for Chemistry, Mainz, Germany (Dutch citizen),
Professor Mario Molina, Department of Earth, Atmospheric and Planetary Sciences and Department of Chemistry, MIT, Cambridge, MA, USA and
Professor F. Sherwood Rowland, Department of Chemistry, University of California, Irvine, CA, USA
for their work in atmospheric chemistry, particularly concerning the formation and decomposition of ozone.
The ozone layer – The Achilles heel of the biosphere
The atmosphere surrounding the earth contains small quantities of ozone – a gas with molecules consisting of three oxygen atoms (O3). If all the ozone in the atmosphere were compressed to a pressure corresponding to that at the earth’s surface, the layer would be only 3 mm thick. But even though ozone occurs in such small quantities, it plays an exceptionally fundamental part in life on earth. This is because ozone, together with ordinary molecular oxygen (O2), is able to absorb the major part of the sun’s ultraviolet radiation and therefore prevent this dangerous radiation from reaching the surface. Without a protective ozone layer in the atmosphere, animals and plants could not exist, at least upon land. It is therefore of the greatest importance to understand the processes that regulate the atmosphere’s ozone content.
Paul Crutzen, Mario Molina and Sherwood Rowland have all made pioneering contributions to explaining how ozone is formed and decomposes through chemical processes in the atmosphere. Most importantly, they have in this way showed how sensitive the ozone layer is to the influence of anthropogenic emissions of certain compounds. The thin ozone layer has proved to be an Achilles heel that may be seriously injured by apparently moderate changes in the composition of the atmosphere. By explaining the chemical mechanisms that affect the thickness of the ozone layer, the three researchers have contributed to our salvation from a global environmental problem that could have catastrophic consequences.
How this knowledge evolved
Ozone is formed in the atmosphere through the splitting of ordinary oxygen molecules (O2) by ultra-violet radiation from the sun. The oxygen atoms thereby liberated react with the molecular oxygen according to:
O2+ uv-light -> 2O
O+O2+M -> O3+M
where M is a random air molecule (N2 or O2).
The English physicist Sidney Chapman formulated in 1930 the first photochemical theory for the formation and decomposition of ozone in the atmosphere. This theory, which describes how sunlight converts the various forms of oxygen from one to another, explains why the highest contents of ozone occur in the layer between 15 and 50 km, termed the ozone layer (Fig. 1). Later measurements, however, showed appreciable deviations from Chapman’s theory. The calculated ozone contents were considerably higher than the observed ones. Thus, there must be other chemical reactions contributing to the reduction of the ozone content. Some years later the Belgian Marcel Nicolet contributed important knowledge of how the decomposition of ozone was enhanced by the presence of the hydrogen radicals OH and HO2.
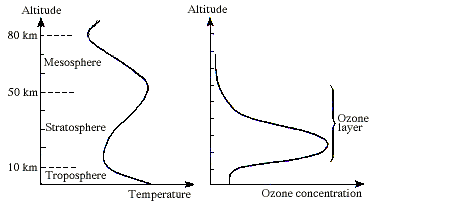
Fig. 1. Variation in temperature and ozone concentration up through the atmosphere
The scientist to take the next fundamental step towards a deeper understanding of the chemistry of the ozone layer was Paul Crutzen. In 1970 he showed that the nitrogen oxides NO and NO2 react catalytically (without themselves being consumed) with ozone, thus accelerating the rate of reduction of the ozone content.
NO + O3 -> NO2 + O2
NO2+O -> NO+O2
O3+uv-light -> O2+O
______________________
Net result: 2O3 -> 3O2
These nitrogen oxides are formed in the atmosphere through the decay of the chemically stable nitrous oxide N2O, which originates from microbiological transformations at the ground. The connection demonstrated by Crutzen between microorganisms in the soil and the thickness of the ozone layer is one of the motives for the recent rapid development of research on global biogeochemical cycles.
The first threat noted: supersonic aircraft
The power of nitrogen oxides to decompose ozone was also noted early by the American researcher Harold Johnston, who carried out extensive laboratory studies of the chemistry of nitrogen compounds. In 1971 he pointed out the possible threat to the ozone layer that the planned fleet of supersonic aircraft and supersonic travel (SST) might represent. These aircraft would be capable of releasing nitrogen oxides right in the middle of the ozone layer at altitudes of 20 km. Crutzen’s and Johnston’s work gave rise to a very intensive debate among researchers as well as among technologists and decision-makers. This was also the start of intensive research into the chemistry of the atmosphere which has made great progress during the past several years. (The subsequent cancellation of plans for a large SST fleet had other reasons than the environmental risks they involved.)
Spray cans and refrigerators damage the ozone layer
The next leap in our knowledge of ozone chemistry was in 1974, when Mario Molina and Sherwood Rowland published their widely noted Nature article on the threat to the ozone layer from chlorofluorocarbon (CFC) gases – “freons” – used in spray bottles, as the cooling medium in refrigerators and elsewhere and plastic foams. Molina and Rowland based their conclusions on two important contributions by other researchers:
– James Lovelock (England) had recently developed a highly sensitive device of measuring extremely low organic gas contents in the atmosphere, the electron capture detector. Using this he could now demonstrate that the exclusively man-made, chemically inert, CFC gases had already spread globally throughout the atmosphere.
– Richard Stolarski and Ralph Cicerone (USA) had shown that free chlorine atoms in the atmosphere can decompose ozone catalytically in similar ways as nitrogen oxides do.
Molina and Rowland realised that the chemically inert CFC could gradually be transported up to the ozone layer, there to be met by such intensive ultra-violet light that they would be separated into their constituents, notably chlorine atoms. They calculated that if human use of CFC gases was to continue at an unaltered rate the ozone layer would be depleted by many percent after some decades. Their prediction created an enormous attention. For the CFC gases were used in many technical processes and their very chemical stability and non-toxicity were thought to render them environmentally ideal. Many were critical of Molina’s and Rowland’s calculations but yet more were seriously concerned by the possibility of a depleted ozone layer. Today we know that they were right in all essentials. It was to turn out that they had even underestimated the risk.
Ozone content over Antarctica
Molina’s and Rowland’s report led to certain restrictions on CFC release during the late 1970s and early 1980s. Not until 1985, when the real shock came, was there any real urgency in the international negotiations on release restrictions. Then the Englishman Joseph Farman and his colleagues noted a drastic depletion of the ozone layer over the Antarctic, the “ozone hole” (Fig. 2). The depletion was, at least periodically, far greater than expected from earlier calculations of the CFC effect. The debate among researchers now intensified. Was this a natural climatic variation or was it chemical decomposition brought about by mankind? Thanks to pioneering research by many researchers, among them Crutzen, Molina and Rowland, as well as Susan Solomon and James Anderson, both from the USA, the picture has now cleared. The depletion is caused chiefly by ozone reacting chemically with chlorine and bromine from industrially manufactured gases.
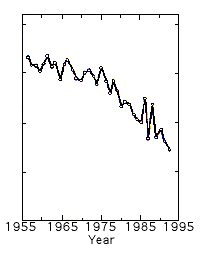 |
| Fig 2. Thickness of the ozone layer (mean monthly value for October) over Halley Bay, Antarctica. Note the drastic depletion since the end of the 1970s. |
The surprisingly rapid depletion of the ozone layer over Antarctica could not be explained by transport processes or by gas phase chemical reactions. An alternative mechanism must exist which could accelerate the decomposition of ozone. Crutzen and colleagues identified this mechanism as chemical reactions on the surface of cloud particles in the stratosphere. Thus, the Antarctic ozone depletion appears to be connected with the extremely low prevailing temperatures, which lead to condensation of water and nitric acid to form “polar stratospheric clouds” (PSCs). The ozone-decomposing chemical reactions are greatly reinforced by the presence of cloud particles. This understanding has led to an exciting new branch of atmospheric chemistry: “heterogeneous” chemical reactions on particle surfaces.
The ozone layer and the climate
The ozone problem also has interesting connections with the issue of how mankind is affecting the climate. Ozone, like carbon dioxide and methane, is a greenhouse gas that contributes to high temperatures at the surface of the earth. (CFC gases have a similar effect). Model calculations have shown that the climate is specially sensitive to changes in the ozone content in the lower layers, the troposphere. Here the ozone content has increased markedly during the past century, chiefly because of the release of nitric oxide, carbon monoxide and gaseous hydrocarbons from vehicles and industrial processes and from the combustion of biomass in the tropics. The elevated ozone content in lower atmospheric layers is itself an environmental problem for the damage it can cause to crops and human health. Paul Crutzen has been the world’s leading researcher in mapping the chemical mechanisms that determine the ozone content at these levels.
What can we expect in the future?
Thanks to our good scientific understanding of the ozone problem – and very largely to Crutzen, Molina and Rowland – it has been possible to make far-reaching decisions on prohibiting the release of gases that destroy ozone. A protocol on the protection of the ozone layer was negotiated under the auspices of the United Nations and signed in Montreal, Canada, in 1987. Under the latest tightening-up of the Montreal Protocol, the most dangerous gases will be totally banned from 1996 (developing countries have a few years’ grace to introduce substitutes that do not harm the ozone layer). Since it takes some time for the ozone-destroying gases to reach the ozone layer we must expect the depletion, not only over Antarctica but also over parts of the Northern Hemisphere, to worsen for some years to come. Given compliance with the prohibitions, the ozone layer should gradually begin to heal after the turn of the century (Fig. 3). Yet it will take at least 100 years before it has fully recovered.
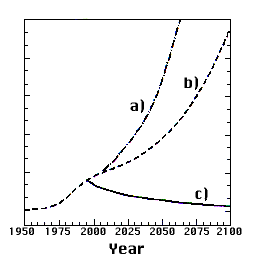
Fig 3. Change in the chlorine content in the stratosphere up to the present and three different future scenarios: a) Without restrictions on release, b) Limitations according to the original Montreal Protocol of 1987 c) The release limitations now internationally agreed. (Chlorine content is a measure of the magnitude of ozone depletion.)
| Further reading |
| Dotto, L. and Schiff, H. I., The ozone war, Doubleday, Garden City, New York, 1978. |
| Graedel, T. E. and Crutzen, P.J., Atmosphere, climate and change. Scientific American Library, 1995. |
| Gribbin, J., The hole in the sky, Corgi Books, London, 1988. |
| Ozonskiktet, Faktablad från Naturvårdsverket, Februari 1992. |
| Rodhe, H. och Bolin, B., Luftföroreningar, I Tidens miljöbok – En forskarantologi, Tidens förlag, 1992. |
| Rowland, F. S. and Molina, M. J., Ozone depletion: 20 years after the alarm, Chemical and Engineering News 72, 8-13, 1994. |
| Scientific assessment of ozone depletion 1994, WMO Report 37, World Meteorological Organization and United Nations Environment Programme, Geneva, 1995. |
| Toon, O. B. and Turco, R. P., Polar stratospheric clouds and ozone depletion, Scientific American 264, 68-74, 1991. |
| Wayne, R. P. Chemistry of Atmospheres, Oxford Science Publications, 1993. |
| WMO and the ozone issue, World Meteorological Organization, Report 778, 1992. |
Paul Crutzen was born in 1933 in Amsterdam. Dutch citizen. Doctor’s degree in meteorology, Stockholm University, 1973. Member of the Royal Swedish Academy of Sciences, the Royal Swedish Academy of Engineering Sciences and Academia Europaea.
Professor Paul Crutzen
Max-Planck-Institute for Chemistry
P.O. Box 3060
D-55020 Mainz, Germany.
Mario Molina was born in 1943 in Mexico City, Mexico. PhD in physical chemistry, University of California, Berkeley. Member of the US National Academy of Sciences.
Professor Mario Molina
Department of Earth, Atmospheric and Planetary Sciences
MIT 54 – 1312
Cambridge MA 02139, USA
F. Sherwood Rowland was born in Delaware, Ohio, USA, 1927. Doctor’s degree in chemistry, University of Chicago, 1952. Member of the American Academy of Arts and Sciences and of the US National Academy of Sciences, where he is currently Foreign Secretary.
Professor F. Sherwood Rowland
Department of Chemistry
University of California
Irvine CA 92717, USA
Nobel Prizes and laureates
Six prizes were awarded for achievements that have conferred the greatest benefit to humankind. The 14 laureates' work and discoveries range from quantum tunnelling to promoting democratic rights.
See them all presented here.
