The Nobel Prize in Chemistry 1995
 |
||
|
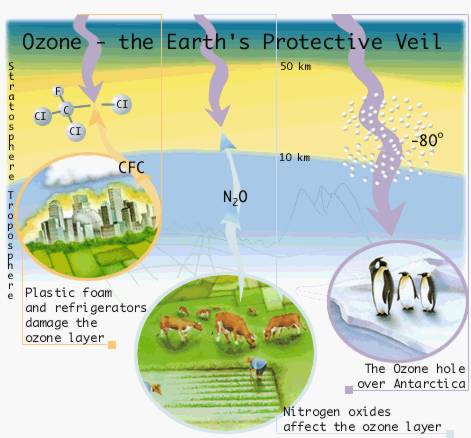
Humans affect the earth’s environment by releasing substances that deplete the protective ozone layer. The Nobel laureates in Chemistry 1995, Mario Molina, Sherwood Rowland and Paul Crutzen, clarified the mechanisms for the chemical reactions involved. The results have led to extensive limitations on the release of ozone-damaging substances. Read more about the ozone layer and:
|
Ozone (03) is formed in the stratosphere through the splitting of ordinary oxygen molecules (02) by ultraviolet radiation from the sun. The liberated oxygen atoms (O) react, through the mediation of some arbitrary molecule (M), with molecular oxygen as follows: 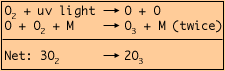 Ozone in small amounts Ozone for better and for worse |
Nobel Prizes and laureates
Six prizes were awarded for achievements that have conferred the greatest benefit to humankind. The 14 laureates' work and discoveries range from quantum tunnelling to promoting democratic rights.
See them all presented here.
